SQF Edition 10.0: What’s Changing and What to Do Now


SQF Edition 10.0: What’s Changing and What to Do Now
March 26, 2026
7
min read
Guide to FDA 21 CFR Part 11: Electronic Records, e-Signatures and Audit-Ready Compliance


Guide to FDA 21 CFR Part 11: Electronic Records, e-Signatures and Audit-Ready Compliance
March 04, 2026
8
min read
FDA QMSR Inspection Readiness for 2026 and Beyond


FDA QMSR Inspection Readiness for 2026 and Beyond
December 11, 2025
5
min read
ISO 9001:2026 Transition Roadmap and QMS Guide
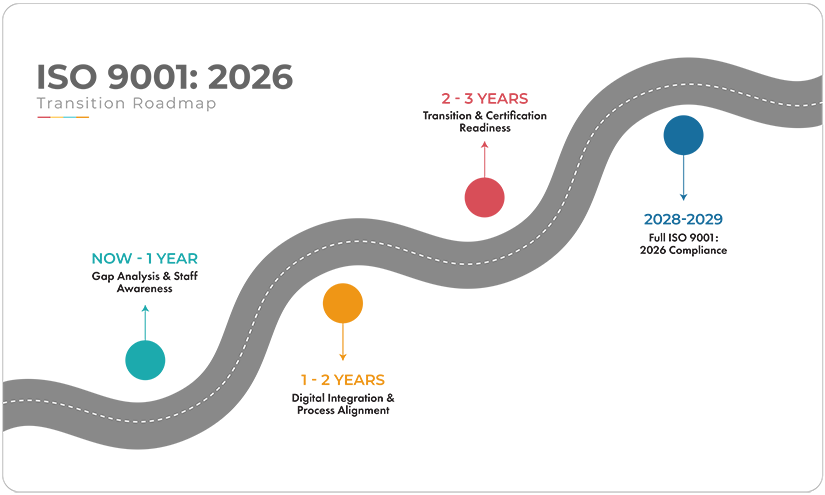

ISO 9001:2026 Transition Roadmap and QMS Guide
November 06, 2025
7
min read