Manage Quality Events with Ease
Capture, track and resolve quality events to improve compliance and reduce risk.
Capture Quality Events
Log deviations, incidents and nonconformances with ease.
Secure Event Database
Store all event details in one searchable, compliant hub.
Auto Risk Prioritization
Score events automatically to highlight the most critical.
Fast CAPA Launch
Trigger corrective actions instantly from quality incidents.
Regulatory Confidence
Maintain full traceability to meet ISO, FDA and GMP standards.
Analyze Event Trends
Use dashboards to spot patterns and prevent repeat issues.
What is quality event management software?
Quality event management is the process of capturing, assessing and resolving unexpected quality issues, such as deviations, incidents, and process failures, that can impact product safety, compliance or operational performance. This process is essential in regulated industries like life sciences, manufacturing, and food and beverage production, where ISO 9001, FDA 21 CFR Part 11 and GMP standards require organizations to document and address quality-related events promptly and consistently.
QT9’s quality events software gives organizations a centralized system to track and manage all quality events in real time. From initial event logging and risk scoring to corrective actions and approvals, every step is streamlined and traceable. Fully integrated with the QT9 QMS, it connects seamlessly with modules like CAPA, audits and customer complaints, helping businesses improve compliance, reduce risk and drive continuous improvement across operations.
Interactive Video Demo

QT9 QMS Quality Events Demo
QT9 QMS is a fully-validated platform
We do all the software validation for you. No extra charge.
Centralize quality events
-
Closed-loop quality event management: Create, track and trend quality event investigations and outcomes in one place.
-
Connect quality events: Quality Events serves as your central hub to quality issues that can create corrective actions, nonconforming products, customer feedback and even deviations.
-
Eliminate data silos: Work together in one system for accurate traceability and compliance.
-
Manage custom fields: Create unlimited user-defined fields and save your best practices for the future.
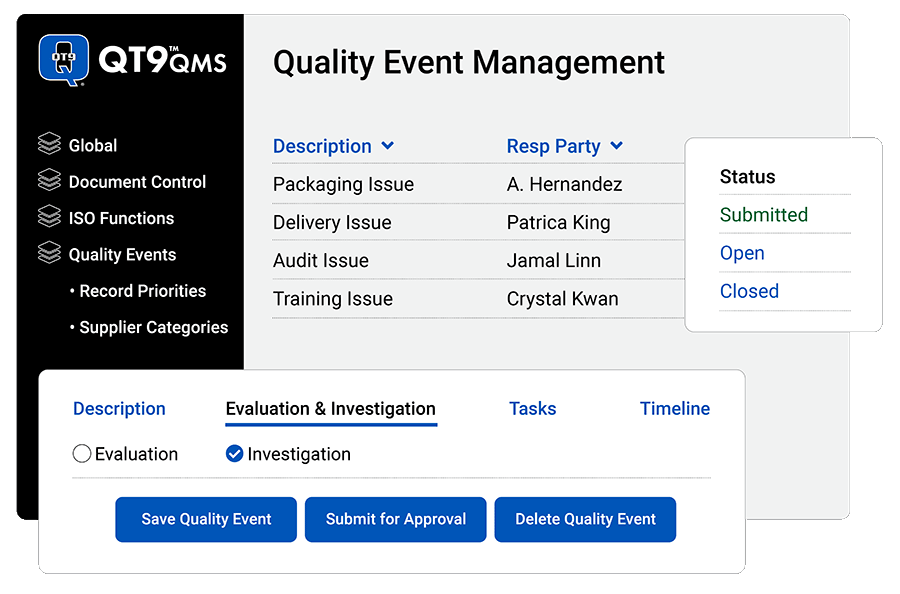
Capture end-to-end quality event management
-
Record any event affecting your quality system: Enable your team to investigate, delegate tasks, attach files, tag types and categories for trend analysis, and link to other modules in QT9 QMS.
-
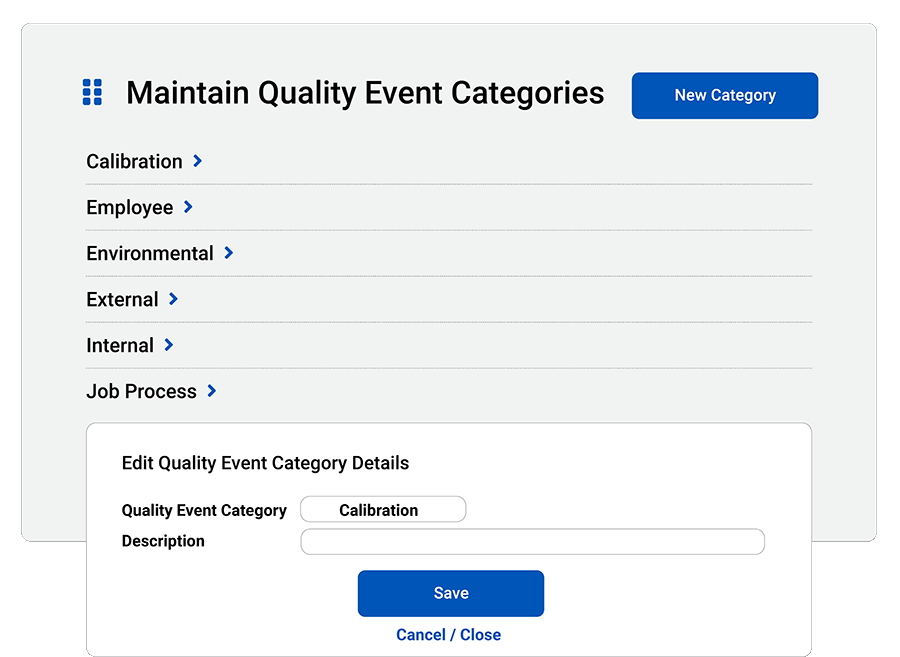
Assign categories: Organize quality events with your own types, categories and dispositions.
-
Manage workflows: Route action items to the right person with automated reminders.
-
Standardize quality event processes: Consistently track quality events the same way to ensure no information gets missed.
Track and trend quality events
-
Real-time reporting: Keep pace with trends in quality events for better decision-making.
-
See quality events your way: Make it easy to see the most relevant information for your team by creating multiple custom views within quality events.
-
Stay on track: Reduce delays with automation efficiencies, such as email alerts and electronic signature approvals.
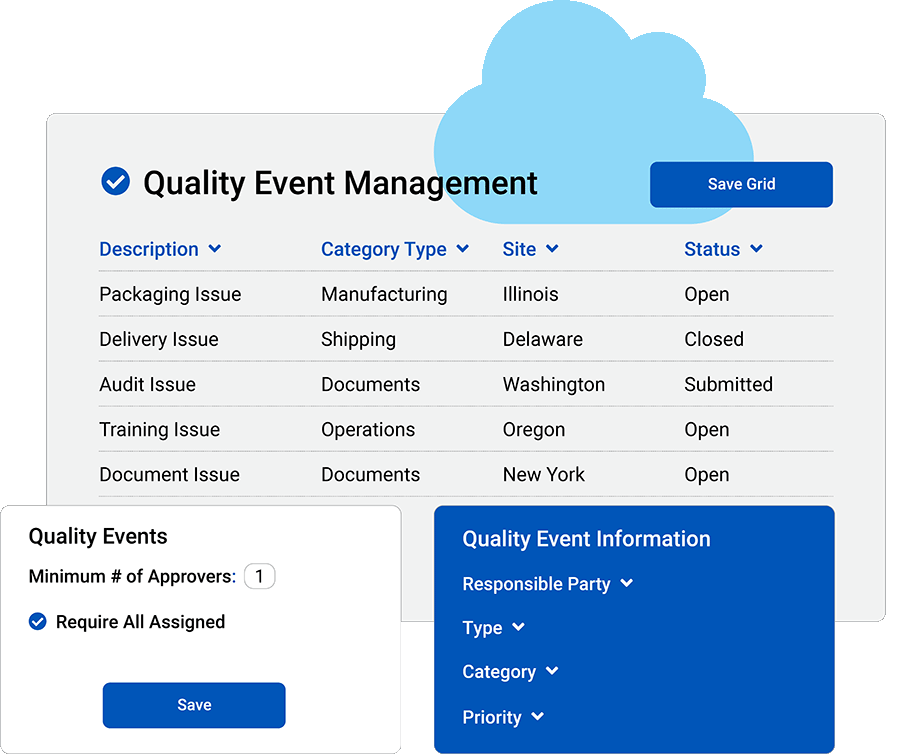
Integrate your quality events
Create a shared hub for collaboration.
Improve Collaboration
Link quality events to customers, suppliers, processes and products.
Manage Tasks
Assign responsibilities, set deadlines and track progress.
Responsible Party
Pre-assign approvers and create responsible parties.
Improve Traceability
See a timeline of every action along with lot and serial traceability.
Get Real-Time Analytics
Ensure Compliance
Show proof of proper handling, supporting FDA Current Good Manufacturing Practices (cGMP).
Initiate Investigations
Launch and explore evaluations and investigations. Assign dispositions and track verification notes.
Link Files
Tackle quality events together by sharing files attachments to keep everyone on the same page.
See what quality leaders say about QT9 QMS
Get end-to-end quality management
FAQ: Quality Events
A quality event is part of a quality management system used to identify any departure from an expected behavior that could effect quality. This could be anything from an incorrect temperature recording to a customer complaint.
Every quality event is different, but having a standardized process to follow helps ensure no task is overlooked. The general framework to address a quality event involves collecting basic information about the incident, assessing level of risk, initiating an investigation if so warranted, and documenting each step for compliance and future trend analysis. The QT9 QMS Quality Events Module standardizes the process and automatically documents processes for easier tracking and traceability.
Related quality management resources

How CAPA Backlogs Drive the Cost of Poor Quality, Risk and Operational Drag

EUDAMED 2026: Is Med-Tech Ready for Full MDR Enforcement?

SQF Edition 10.0: What’s Changing and What to Do Now
Try QT9 for free
Ready to simplify your quality processes? No credit card needed.