ERP that Connects Operations, Quality and Compliance
Built for regulated manufacturers that demand audit-ready traceability, operational control and pre-validated workflows for FDA, ISO and global standards.
ISO 9001 | ISO 13485 | 21 CFR Part 11
Cloud, On-Prem or Hybrid
Go live in months, not years
US-based support + guided onboarding
Built for Regulated Manufacturing Industries
Select your industry to see how QT9 ERP supports your compliance and operational needs.
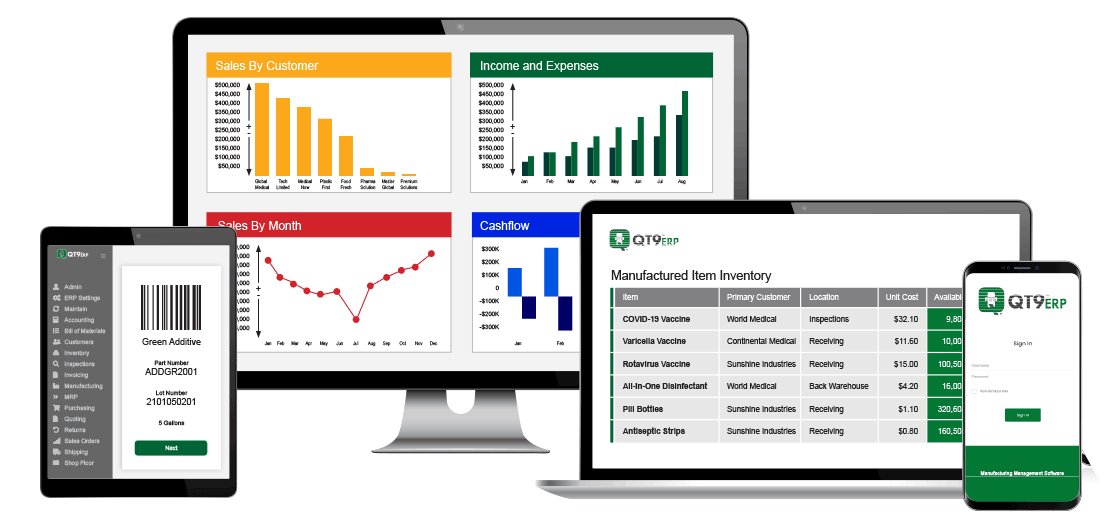
Next-Generation ERP Automation for Regulated Manufacturers
Get benefits beyond everyday automations with embedded workflows that enable you to work smarter and faster. Eliminate time-consuming manual manufacturing processes and gain real-time insights to reduce human errors and speed decision-making.
Pre-Validated ERP Software
QT9 ERP provides a pre-validated environment, enabling faster implementation and increased return on investment. Every module and global feature goes through a step-by-step validation eliminating the need to invest in it yourself, saving your time, your money and your resources.
End-to-End Traceability
Track every part, product and process in real time with full forward and backward traceability. QT9 ERP gives manufacturers complete visibility into raw materials, inventory movements, production history and finished goods. This ensures quick recalls, simplified audits and compliance with regulations like ISO 9001 and FDA 21 CFR. From purchase order to shipment, QT9 ERP makes it easy to trace data across the supply chain and drive accountability at every stage.
Customer, Supplier and Compliance Portals
QT9 ERP customer, supplier and compliance web portals ensure secure access to vital documents, and improve communication and collaboration.
Faster Issue Detection and Resolution
QT9 ERP enables early identification of issues so you can take immediate actions to minimize impact. This helps prevents defects, reduces downtime and minimizes compliance risks.
Reduce the Learning Curve
From startups to enterprises, get up to speed with a platform that’s quick to learn and easy to love. QT9 ERP helps teams minimize mistakes in data entry and workflows, ensuring accurate production timelines, delivery dates and seamless business processes.
ERP Software that Connects Your Entire Business
Everything you need to run regulated manufacturing, fully integrated in one ERP platform.
MRP Planning
Automatically generate work orders and purchase orders based on real-time demand.
Inventory Management
Track inventory levels, movements and locations with full traceability.
Purchasing Automation
Create and manage purchase orders, supplier data and receipts all in one place.
Shop Floor Control
Schedule jobs, assign tasks and monitor production progress in real time.
Invoicing & Billing
Easily generate, send and track invoices with built-in status monitoring.
Shipping & Receiving
Manage shipments, print labels and log receipts to keep logistics on track.
Customer Management
Track customer interactions, sales history and communication in one view.
Supplier Management
Manage supplier records, contacts and performance with full audit history.
Custom Reporting
Generate reports using live ERP data and export to Excel or BI tools.
Everything You Need to Run Manufacturing in One ERP Platform
Modern ERP Software Built for
Growing Businesses
From automation and dashboards to multi-site visibility, QT9 ERP connects your entire operation in one easy-to-use, scalable platform
Directly link data between modules. Whether you are creating purchase orders from MRP planning, scheduling jobs to your shop floor manager or receiving inventory from PO inspection, QT9 ERP's automation will keep you working as efficiently as possible.
See the big picture. QT9 ERP keeps you up to date on your past, present and future business activities. Easily spot gaps and overlaps in your operation and quickly make adjustments utilizing real-time dashboards and built-in analytics.
Tie it all together and eliminate the need for switching between systems by keeping everything organized in one place. QT9 ERP allows you to share data across various sites and functions from a single platform. With true multi-site capability, QT9 ERP can be the umbrella that keeps your organization centralized.
Do more with less. QT9 ERP is easy to use. Quickly automate manual, redundant, time-consuming tasks. Minimize the hassle of duplicate data entry and reduce the risk for errors in the process with QT9's user-friendly solution.
All modules are included with QT9 ERP. As business needs increase, you can quickly shift from module to module so you're sure to use those that have the biggest impact on efficiency and growth.


Pre-Validated ERP Software
QT9 ERP is pre-validated for accelerated compliance.
-
FDA and ISO compliance out-of-the-box
-
Rapid cloud deployment for faster implementation
-
Audit-ready with timeline traceability
-
Electronic signature approvals and secure access controls
-
Continuous validation and updates
|
• ISO 13485 |
• FDA 21 CFR Part 11 |
QT9 ERP includes a complete execution of all protocols, including Installation Qualification (IQ), Operation Qualification (OQ) and Performance Qualification (PQ).
-
Eliminates costly internal ERP validation efforts that can take months of staff time or cost thousands in consultant fees.
-
Reduces downtime waiting for your ERP to be approved for use.
-
Minimizes the risk of any failed audits due to flawed validation protocols.
ERP Compliance Portal for Regulated Industries
Digitize device history records (DHR/MDR) and batch records (MBR/EBR) in one secure system. Trusted by medical device and pharmaceutical manufacturers across the U.S. and worldwide.
-
Authorized auditors, inspectors, or compliance officers can securely log in to view or print compliance data — including MDR, DHR, MBR and EBR — without exposing unrelated ERP modules.
-
This isolated audit view minimizes compliance risks, safeguards sensitive operational data and ensures faster inspections.
-
Automatically compiles all required components — BOMs, routings, QMS documents, inspection data, job travelers and approvals — into one validated report.
-
Eliminates manual data collection, reduces human error and saves manufacturers valuable time in preparing device and batch records.
-
Seamlessly integrates with the QT9 QMS, ensuring MDR/DHR and MBR/EBR always reflect the latest approved documents, inspection plans, and production data.
-
Guarantees only current revisions of controlled documents are included, maintaining full traceability and compliance.
-
Automatically links production jobs, BOMs, materials, inspections, and approvals to each generated record.
-
Helps manufacturers comply with FDA 21 CFR Part 210/211/820, ISO 13485 and GMP requirements.
-
Provides a transparent, auditable trail from design through production, supporting both local and global regulatory demands.
-
Supports single-level and multi-level record generation for complex assemblies or multi-stage production.
-
Ensures visibility across all subassemblies, components, and processes, critical for regulated manufacturing operations.

Get QT9 ERP in the cloud or on-premise.
Flexible deployment to match your IT strategy, without compromising security or speed.
One Platform, Complete Control
QT9™ ERP + QMS brings it all together in a single pre-validated platform. Maintain complete traceability, accelerate audits and stay compliant with FDA 21 CFR Part 11 and ISO 13485 — without the headaches of multiple disconnected tools.
Trusted by leading medical device, biotech and pharmaceutical companies worldwide.
Go Paperless: Streamline Compliance with EBR and eDHR Solutions

Electronic Batch Records
Go paperless by creating Electronic Batch Records (EBR) and Master Batch Records (MBR) with one click using QT9 ERP and QT9 QMS software.

Electronic Device History Records
Go paperless and create electronic Device History Records (eDHR) and Device Master Records (eDMR) with one click using QT9 QMS and QT9 ERP software.
See What Quality Leaders Say About QT9 ERP
FAQs: QT9 ERP
QT9 ERP is Enterprise Resource Planning system designed for manufacturers and regulated industries to manage and connect all business operations, including production, inventory management, purchasing and supplier management, in one platform. It provides audit-ready traceability, operational control and pre-validated workflows that meet FDA, ISO and global standards.
QT9 ERP is built for regulated manufacturing environments and offers native integration with QT9 QMS, enabling seamless data flow between operations and quality. Unlike traditional ERP systems, it combines compliance, traceability and manufacturing execution without requiring third-party integrations.
QT9 ERP comes pre-validated and includes embedded workflows that improve efficiency, eliminate inaccuracies and provide full traceability. QT9 offers flexible deployment options to match your IT strategy, via the cloud or on premise.
Users can automate device history records (DHR/MDR/MDF) and batch records (MBR/EBR) in one secure solution.
Any industry can benefit from using QT9 ERP, however, it is especially helpful for organizations in regulated and quality-driven industries, such as:
- Medical devices
- Pharmaceuticals and life sciences
- Aerospace and defense
- Automotive
- General manufacturing
These industries rely on QT9 ERP to manage production while maintaining strict compliance standards.
QT9 ERP includes a comprehensive set of features to manage operations, including:
- Production and job management
- Inventory and warehouse control
- Purchasing and Material Requirements Planning (MRP)
- Supplier management
- Multi-Level Bill of Materials (BOM)
- Scheduling and capacity planning
- Traceability and lot control
- Reporting and real-time dashboards
All features are integrated within a single system for complete operational visibility.
QT9 ERP integrates natively with QT9 QMS, allowing quality and operational data to function as one system. This enables:
- Real-time linkage between production and quality events
- Automated generation of batch records and device history records
- Immediate visibility into nonconformances, CAPAs and inspections tied to jobs and materials
This unified approach eliminates silos and improves compliance and traceability.
Yes. QT9 ERP is designed to support compliance with standards such as:
- FDA 21 CFR Part 11 and Part 820
- ISO 9001 and ISO 13485
- AS9100 and IATF 16949
It includes built-in audit trails, electronic signatures and full traceability to help organizations meet regulatory requirements.
QT9 ERP provides end-to-end traceability by tracking materials, components and finished goods across the entire production lifecycle. Users can trace products from raw materials through manufacturing to final delivery, supporting recalls, audits and compliance reporting.
Yes. QT9 ERP improves production efficiency by:
- Providing real-time visibility into operations
- Optimizing scheduling and resource allocation
- Identifying bottlenecks and delays early
- Connecting quality events directly to production workflows
This helps teams respond quickly and minimize downtime.
QT9 ERP offers both cloud-based and on-premise deployment options, allowing organizations to choose the setup that best fits their IT infrastructure, security requirements and scalability needs.
Implementation timelines vary depending on complexity, but QT9 ERP is designed for faster deployment compared to traditional ERP systems. Its integrated architecture eliminates the need for extensive customization and third-party integrations.
Yes. QT9 ERP supports multi-site and global operations, enabling organizations to manage multiple facilities, standardize processes and maintain consistent data across locations.
QT9 ERP provides real-time dashboards and reporting tools that give visibility into supply and demand, production, inventory management, supplier data and financials. This allows leaders to make faster, data-driven decisions and proactively address issues before they impact operations.
Yes. QT9 ERP is designed to scale with organizations, supporting increased production volume, additional users and expanded compliance requirements without needing to switch systems.
Manufacturers choose QT9 ERP because it:
- Provides everything needed in one, fully integrated platform
- Seamlessly integrates with QT9 QMS
- Improves traceability and compliance
- Reduces manual processes and data silos
- Enables real-time visibility across operations
- Supports regulated industries without heavy customization
This makes QT9 ERP a great fit for organizations that need both operational control and regulatory confidence.
Explore More ERP Resources

Five Signs You’ve Outgrown Your ERP System

ERP in Supply Chain Management: How Manufacturers Gain Control, Visibility and Predictability

Best Software for Inventory Management in Regulated Manufacturing
Run Your Operation With Clarity and Control
See how QT9 simplifies operations today and scales with you tomorrow.
Schedule Demo
See a personalized demo of QT9 QMS with our sales team.
Start a Trial
Explore how easy QT9 QMS is able to connect operations.
Get a Quote
See how much QT9’s all-in-one platform costs.
Call QT9
Available M-F 8 AM to 5 PM CT. Speak to a QT9 expert.