Streamline Quality and Compliance with QT9 QMS Software
Pre-validated Quality Management Software built to automate audits, CAPA, training and document control in a secure cloud or on-premise platform that scales with your business.
25+ QMS Modules for Complete Quality Management
Introducing The QT9 Q-Cast

Drive Quality Performance Across your Organization
QT9 QMS enables real-time access to quality data, streamlines team communication, and automates alerts to ensure faster, smarter decisions.
Centralize quality data, information and processes in one system for easy accessibility wherever employees are located.
Invest in continuous improvement by providing a scalable, fully-automated quality system that boosts accuracy and efficiency.
Accelerate productivity and get products to market faster by streamlining quality and compliance. Dynamically populated data and automated workflows help keep projects on track.
Get the tools you need to stay agile and more easily respond to changing regulatory and market demands. Let QT9 handle the busy work, while your team focuses on innovation.

An Award-Winning Platform. Loved by Customers.
QT9 QMS is a leader in Quality Management Software on G2 — the largest software marketplace.
82%+ of compliance leaders cite third-party risks as material issues — making integrated compliance tools not just useful, but essential.
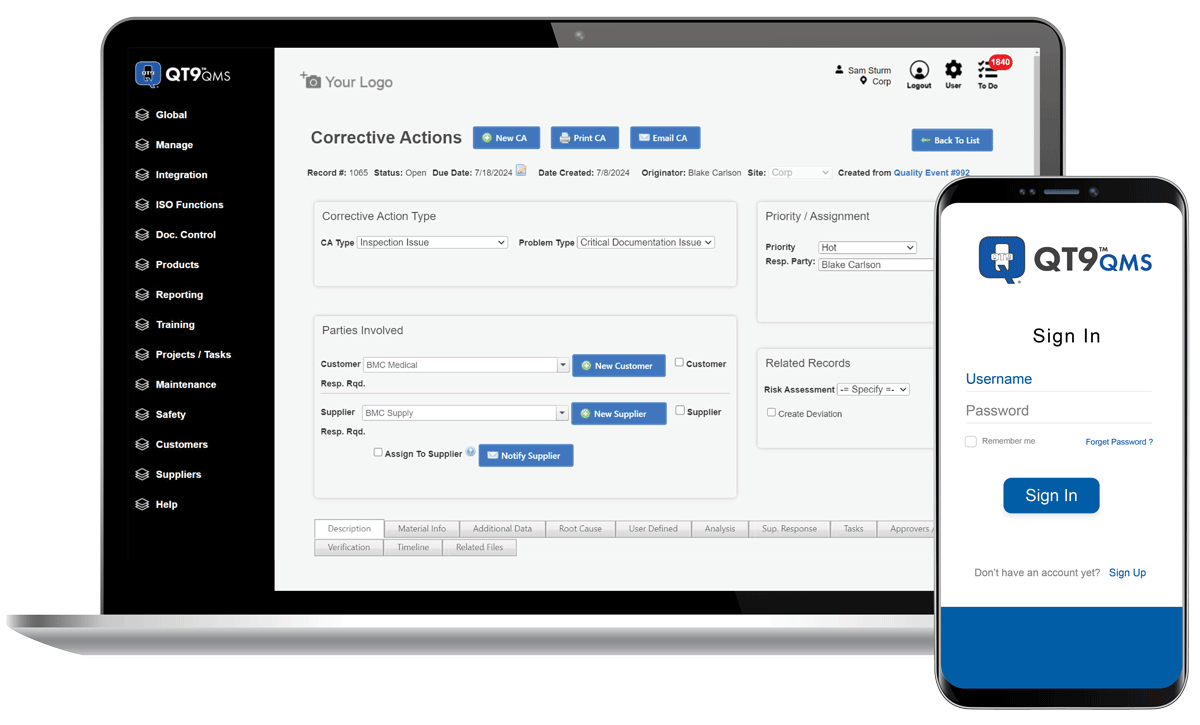
Get QT9 QMS in the Cloud or on Premise.
Flexible deployment to match your IT strategy without compromising security or speed.
QT9 QMS next-generation automation
Get benefits beyond everyday automations with embedded workflows that enable you to work smarter and faster. Eliminate time-consuming manual quality checks and gain real-time insights to reduce human errors and speed decision-making.
QT9 QMS is pre-validated
QT9 QMS provides a pre-validated environment, enabling faster implementation and return on investment. Every module and global feature goes through a step-by-step validation. What's more, you don't have to invest in it yourself.
Provide accurate audit trails for inspections
QT9 QMS provides timeline traceability that gives transparency into every action. This makes it easier to identify defects or trace defective batches for targeted recalls.
Employee, customer and supplier portals
QT9 QMS customer, supplier and employee web portals ensure secure access to vital documents, improving communication and collaboration.
Faster issue detection and resolution
QT9 QMS enables you to identify quality issues as they occur, so you can take immediate corrective actions to minimize impact. This prevents defects, reduces downtime and minimizes compliance risks.
Reduce the learning curve
From startups to enterprises, get up to speed with a platform that’s quick to learn and easy to love. QT9 QMS helps teams minimize mistakes in data entry and workflows, ensuring accurate documentation, seamless audits and compliance with industry standards.
Pre-Validated QMS for ISO, FDA, EU MDR & AS9100
Simplified international medical device quality compliance.

EU MDR Compliance, Simplified

Built-in electronic documentation compliance with full traceability

End-to-end U.S. pharmaceutical compliance

Simplified compliance for medical devices in the U.S.

A one-stop solution for meeting ISO 9001 requirements

A streamlined environmental management system

Streamline and standardize testing lab compliance.

Accurate and efficient aerospace and defense compliance

Centralize and simplify automotive IATF 16949 compliance

Meet food safety regulations with accuracy and efficiency

Food safety compliance throughout the supply chain

QMS Features that give your Team a Competitive Edge
Expand QT9 QMS with built-in modules designed to fit your unique workflows.
Centralize Documents
Access documents in one place for better document management.
Streamline Audits
Track and manage scheduling details and the results of your audits.
Monitor Deviations
Easily track your temporary deviations with email alerts and to-do lists.
Minimize Risk
Manage risk analysis by assigning risk score, tasks, priorities, approvals, etc.
Modernize Inspections
Capture inspection details online and link related documents for reference.
Track Engineering Changes
Control ECR/ECN by assigning tasks, email alerts and electronic approvals.
Train Employees
Manage training online for employees and job roles with personalized tests.
Not Ready for a Demo? Start Here.
Three tools to help you build the case, benchmark your readiness, and find the right fit.
Prefer to explore on your own Try QT9 free — no credit card required →

Pre-Validated QMS Software
QT9 QMS is pre-validated for accelerated compliance.
-
FDA and ISO compliance out-of-the-box
-
Rapid cloud deployment for faster implementation
-
Audit-ready with timeline traceability
-
Electronic signature approvals and secure access controls
-
Continuous validation and updates
|
• ISO 13485 |
• FDA 21 CFR Part 11 |
QT9 QMS includes a complete execution of all protocols, including Installation Qualification (IQ), Operation Qualification (OQ) and Performance Qualification (PQ).
-
Eliminates costly internal QMS validation efforts that can take months of staff time or cost thousands in consultant fees.
-
Reduces downtime waiting for your QMS to be approved for use.
-
Minimizes the risk of any failed audits due to flawed validation protocols.
Why Teams Choose QT9 QMS Software
No hidden fees. No user limits. No compromises. See how QT9 QMS by QT9 Software compares to traditional quality management software.
21 CFR Part 11 Compliant
Built-in electronic signature and audit trail compliance for FDA-regulated industries.
QT9 University
Online, on-demand training resources and community forum for continuous learning.
Ready to see the QT9 difference?
Join 1,200+ companies worldwide who trust QT9 QMS software for their quality management.
Celebrated by Experts. Loved by Customers.
QT9 QMS is a leader in Quality Management Software on Capterra, Software Advice and GetApp.
FAQs: QT9 QMS
QT9 QMS is an all-in-one, pre-validated electronic quality management system that enables regulated organizations to automate compliance efforts, centralize quality processes and maintain audit readiness. Available in the cloud or on-premise, it’s 25+ modules offer end-to-end quality management across the product lifecycle.
QT9 QMS is built for regulated manufacturing environments and offers native integration with QT9 ERP, enabling seamless data flow between quality and operations.
QT9 QMS comes pre-validated for easy compliance with standards like FDA 21 CFR Part 11, ISO 9001 and AS9100. It includes IQ, OQ and PQ validation protocols, enabling faster implementation and reduced validation effort.
QT9 QMS provides all 25+ integrated modules out-of-the box, including document control, CAPA, audit management, employee training and risk management. Available in cloud-based or on-premise delivery, QT9 QMS offers flexible deployment, integrated customer, supplier and employee web portals, plus real-time data to enhance business decisions.
Users can automate device history records (DHR/MDR/MDF) and batch records (MBR/EBR) in one secure solution.
QT9 QMS is designed for industries that must meet strict regulatory and quality requirements, such as:
- Medical devices
- Pharmaceuticals and life sciences
- Aerospace and automotive
- General manufacturing
- Food and beverage processing
QT9 QMS provides a full suite of more than 25 integrated quality management applications and features, including:
- Centralized document control with revision tracking
- CAPA and non-conformance management
- Audit management and scheduling
- Training and certification tracking
- Risk management and change control
- Supplier quality management
- Product design and engineering change controls
- Dynamic reporting for full traceability and visibility
- Electronic signature approvals
IQ/OQ/PQ validation
QT9 QMS simplifies compliance by offering:
- Audit management module for continuous audit readiness
- Electronic signatures and built-in audit trails
- Automated workflows and approvals
- Built-in validation protocols (IQ, OQ, PQ)
- Real-time traceability across quality processes
This ensures organizations stay continuously audit-ready and aligned with global regulations.
QT9 QMS supports major global quality regulations and standards, including:
- FDA 21 CFR parts 11, 210, 211 and 820
- ISO 9001 and ISO 13485
- AS9100 and IATF 16949
- EU MDR and GMP frameworks
QT9 QMS can be implemented in as little as 30 days due to its pre-validated environment and ready-to-use modules, significantly reducing deployment time compared to traditional QMS systems. Implementation time varies, however, depending on several factors, including company size and complexity, current processes and procedures, and the number of internal resources dedicated to implementation.
QT9 QMS comes pre-validated with Installation Qualification (IQ), Operational Qualification (OQ) and Performance Qualification (PQ) protocols already completed, reducing internal validation effort and accelerating go-live.
Both. QT9 QMS offers flexible deployment options, including both cloud-based and on-premise environments, allowing organizations to align with their IT, security and compliance requirements.
QT9 QMS improves performance by:
- Automating quality workflows and reducing manual work
- Providing real-time visibility into quality data
- Identifying issues early and triggering corrective actions
- Minimizing errors, defects and compliance risks
This leads to faster decision-making, improved product quality and reduced operational disruption.
Yes. QT9 QMS integrates with ERP systems—but more importantly, it offers a native, built-in integration with QT9 ERP, creating a single, unified platform for quality, manufacturing and business operations.
Unlike typical QMS tools that rely on APIs or middleware, QT9 QMS and QT9 ERP were developed together on a shared architecture. This allows data to flow seamlessly between systems in real time, without synchronization delays, integration failures or duplicate data entry.
Absolutely. QT9 QMS is designed to scale from small teams to global enterprises, supporting multi-site operations, increasing data volumes and expanding compliance requirements without added complexity.
Yes, QT9 QMS has won awards for being user friendly! Its user-friendly interface makes it easy for teams to adapt quickly. Licensing also includes training resources and support to ensure successful onboarding and long-term use.
See what Quality Leaders say about QT9 QMS
Quality Management Resources

Product Rework: The Profit Killer Hiding in Plain Sight

Top Document Control Audit Findings and How to Avoid Them

Always Audit Ready: The New Standard for FDA-Regulated Manufacturers
Try QT9 for Free
Ready to simplify your quality processes? No credit card needed.
Schedule Demo
See a personalized demo of QT9 QMS with our sales team.
Start a Trial
Explore how easy QT9 QMS is able to connect operations.
Get a Quote
See how much QT9’s all-in-one platform costs.
Call QT9
Available M-F 8 AM to 5 PM CT. Speak to a QT9 expert.